2026 North American Sorption Symposium
Tomorrow’s Breakthroughs, Powered by Sorption Science
Bringing the Sorption Science Community Together
Join leading scientists, industry professionals, and academic researchers at the 2026 North American Sorption Symposium, a premier event dedicated to advancing the field of sorption science and materials characterization.
May 19th - 20th, 2026
USDA Forestry Laboratory on the University of Wisconsin–Madison campus.
Madison, Wisconsin

Over two days, attendees will engage in high-level discussions, technical presentations, and collaborative networking sessions that explore the latest breakthroughs in:
- Dynamic Vapor Sorption (DVS)
- Inverse Gas Chromatography (iGC)
-
Breakthrough Analysis
- Applications in clean energy, pharmaceuticals, sustainable materials, porous solids, and more
Whether you specialize in advanced materials, gas separation, pharmaceutical formulation, or environmental research, this symposium offers a unique opportunity to explore how sorption techniques are enabling tomorrow’s scientific and industrial innovations.
Advancing Sorption Science Through Collaboration
The North American Sorption Science Symposium brings together researchers, engineers, and innovators dedicated to exploring adsorption and sorption research across diverse industries. Established to foster dialogue between academia and industry, the symposium provides a platform for sharing insights, exchanging techniques, and forming new partnerships that drive the field forward.
Event Partner:
USDA Forest Products Laboratory in Madison, Wisconsin
Nestled in the University of Wisconsin–Madison campus, the USDA Forest Products Laboratory, offers a welcoming setting for innovation and collaboration. The laboratory has long been a hub for materials research, making it the ideal venue for the 2026 Sorption Symposium.

Event Partner:
USDA Forest Products Laboratory in Madison, Wisconsin
Nestled in the University of Wisconsin–Madison campus, the USDA Forest Products Laboratory, offers a welcoming setting for innovation and collaboration. The laboratory has long been a hub for materials research, making it the ideal venue for the 2026 Sorption Symposium.

Your Guide to the 2026 Sorption Symposium

The North American Sorption Science Symposium is a two day event that brings together researchers, engineers, and innovators dedicated to exploring sorption science across diverse industries.
Established to foster dialogue between academia and industry, the symposium provides a platform for sharing insights, exchanging techniques, and forming new partnerships that drive the field forward.
The Details
The who, where, and why of sorption’s biggest event.
- Academic and industry researchers working with sorption, adsorption, or permeation techniques
- Scientists and lab managers using DVS, iGC, or breakthrough analysis instruments
- Product developers, process engineers, and decision-makers interested in material characterization
- Date: May 19th - 20th, 2026
- Location: Madison, Wisconsin
- Venue: USDA Forestry Laboratory on the University of Wisconsin–Madison campus.
- Hear from expert speakers at the forefront of adsorption science and instrumentation.
- Discover new applications of sorption technology in in pharmaceuticals, energy, and advanced materials.
- Network with peers from across academia, government, and industry.
- Tour the USDA Forest Products Laboratory, a renowned U.S. research facility working on advanced material sustainability.
2026 NA Sorption Symposium Program
The 2026 program features oral presentations and discussion panels covering a broad spectrum of sorption-related research. Attendees can expect sessions focused on DVS, iGC, and breakthrough analysis applications in pharmaceuticals, clean energy, porous materials, and environmental systems.
May 19th, 2026: Day 1 Schedule
A welcome from Dr. Daniel Burnett, Vice President at Surface Measurement Systems to kick off the program.
Coauthors:
Samuel V. Glass (Forest Products Laboratory)
Natalia Farkas (Forest Products Laboratory)
Abstract:
Water vapor sorption isotherms are important for predicting the properties of wood and wood products. However, the slow and complex kinetics of vapor sorption in wood make the use of dynamic vapor sorption (DVS) tools challenging. For example, collecting a full absorption isotherm on a 20 mg sample could take as long as three months if each 10% relative humidity step is held until equilibrium is obtained using a rigorous mass stability criterion for equilibrium. Luckily, the errors from shortening measurement time are systematic, suggesting it may be possible to develop corrections that could be applied to measurements to shorten measurement times. To address this problem, we conducted a worldwide interlaboratory study of water vapor absorption isotherms in wood specimens cut from the same board, where different laboratories took measurements to equilibrium at a broad range of relative humidity steps. Based upon these measurements, we developed a method for correcting data with the commonly used 0.002% min-1stop criterion. This correction method reduces the mean absolute error of sorption data (for the material used in this interlaboratory study) acquired with short hold times to less than 0.001 g g-1.
Abstract:
Sorbents underpin a wide range of technologies, from air and gas purification and carbon capture to atmospheric water harvesting and thermal energy storage. Yet sorbent screening and process design still rely heavily on stepwise isotherm measurements that require long equilibration at each partial pressure, consume costly instrument time, and can obscure true performance when materials drift or degrade during extended runs.
Here we present a transient-response framework that reconstructs both equilibrium uptake and kinetics from short, non-equilibrium experiments. Instead of discrete pressure steps, we use a fast, continuous adsorption-desorption protocol that ramps the relative pressure over a broad range, capturing high-resolution mass-uptake dynamics. We then solve the inverse problem with physics-guided learning: mechanistic transport and rate equations constrain the fit, while Kolmogorov Arnold network-based ordinary differential equation (KAN-ODE) models provide a compact and interpretable representation that can generalize across conditions and separate kinetic and equilibrium contributions.
Across representative sorbents, including hygroscopic desiccants and carbon-based sorbents for organic vapors, this approach recovers full isotherms and effective kinetic parameters with an order-of-magnitude reduction in characterization time, with expected accelerations up to about thirty-fold. Beyond faster screening, learning from dynamic data enables continuous, in situ updates of equilibrium and kinetic parameters for real-time control, setpoint optimization, and drift detection in operating devices.
Finally, we extend the same transient, physics-constrained identification to mixed-gas experiments, allowing simultaneous inference even when an inert carrier gas is present, and towards a multi-species equilibrium and kinetics from coupled responses. The broader message is that steady state is no longer a prerequisite: any solid–gas system that can be excited with a controlled transient and measured in time can be characterized in this way. This opens a path beyond sorbents to reactive gas–solid processes such as redox-active oxides and solid-state gas sensors, where kinetics and material evolution are central and traditional stepwise protocols are too slow or misleading.
Abstract:
Sorbent-based atmospheric water harvesting has attracted interest as a potential pathway to produce freshwater from moisture passively, without requiring centralized infrastructure, and even in arid conditions. However, low productivity and high cost of water severely limit its deployment. Here, we discuss our work simultaneously co-designing moisture sorbents and water production devices guided by physics-based thermodynamic and transport models and technoeconomic analyses towards minimized water cost.
Firstly, we developed and validated through experimental characterization thermodynamic and transport models that predict water uptake, kinetics, and desorption enthalpy of hydrogel-salt sorbents based on their chemical composition and environmental composition. Secondly, we used these models to develop inexpensive hydrogel-salt sorbents capable of capturing ~2 kg of water/kg of material, even in arid conditions (~30% RH), exceeding previous leakage-free sorbents. Thirdly, we integrated these materials into a passive device that was field-tested in the Atacama Desert. Through simultaneous material and device heat and mass transport optimization, we achieved ~1 L/m2/day water production in the desert and ~2 L/m2/day in Boston. Lastly, we developed strategies for material durability to achieve ~200 absorption-desorption cycles and material lifetime exceeding 8 months.
Through our combination of thermodynamic and transport modeling, synthesis, characterization, and device-level optimization, we demonstrate the potential of sorption to address a major sustainability challenge. This motivates our ongoing efforts using sorption for carbon capture, building energy efficiency, and in biomass as a sustainable feedstock.
Enjoy a quick refreshment break.
Abstract:
Understanding moisture sorption mechanisms in biomass-derived carbons is critical for their deployment in water harvesting and environmental adsorption technologies. This presentation examines the evolution of water vapor sorption behavior from raw chestnut shell biomass through biochar and activated carbons produced at 600 °C and 800 °C using dynamic vapor sorption. Experimental isotherms reveal pronounced changes in uptake capacity, hysteresis, and adsorption kinetics with increasing thermal input, reflecting progressive aromatization and pore structure development. The sorption data are quantitatively analyzed using the DS-4 hysteresis model and isotherm fitting approaches reported in the literature, enabling extraction of key parameters governing adsorption–desorption asymmetry and pore filling behavior. Temperature-dependent measurements further allow estimation of the isosteric heat of adsorption, providing thermodynamic insight into water–surface interactions across the material series. Together, these results establish clear structure–property–sorption relationships that guide rational design of sustainable activated carbons for moisture-driven energy and water applications.
Abstract:
Structured Sorbent Sheets remove carbon dioxide (CO2) from the air using Direct Air Capture (DAC) processes. These laminates are composite materials which allow air to flow between gaps of adjacent sheets, in a parallel-sheet contactor format, enabling greater control of pressure drop and capture efficiency compared to traditional packed beds or trays of sorbent beads or pellets.
DAC processes are designed to operate year-round, pulling CO2 from the ambient air. For this reason, it’s critical to understand the performance of Structured Sorbent Sheets across a range of temperatures and humidities. Using a DVS Carbon, simple gravimetric tests can investigate a variety of temperatures and humidities with minimal sample requirements. The methods can be deployed on a variety of samples, and the data collected can inform researchers about a material’s suitability for DAC in various environments.
In this session, we discuss the performance of a 3M Structured Sorbent Sheet in different environments, and we consider the effects of degradation and breakthrough behavior when the material is designed into a DAC process.
Enjoy lunch and also check out our poster presentations.
Posters:
TBD
Abstract:
TBD
Abstract:
Detailed analyses of the drying/desorption of moisture from 3A molecular sieves at temperatures approaching ambient do not exist. Regeneration/activation of sieves is generally performed at temperatures ≥ 220°C, as only temperatures > 270°C are reported to result in irreversible damage to the zeolite and a loss of adsorption capacity. While high activation temperature is ideal for complete desorption and provides the fastest kinetics, drying temperatures above 200°C can cause undesirable “damage” when molecular sieves are bound together by an organic matrix such as an epoxy. Many epoxies can survive brief exposure to temperatures above 200°C, but they chemically degrade over prolonged time periods under these conditions, typically through chain scission processes that release volatile small molecules. For epoxy bound desiccants, mass loss associated with epoxy degradation gets convoluted with the mass loss due to moisture release during high temperature activation processes. In this work, we demonstrate the means to dry 3A molecular sieves, both in powder form and dispersed in an epoxy matrix, at much lower temperatures. In the process, we identify the time and energy required to remove the moisture content. The implications of drying temperature on epoxy mass are also quantified and illustrate the impact epoxy mass loss has on the ability to use mass measurements to validate the activation state of epoxy bound desiccants.
Abstract:
Polymer membranes that selectively regulate small molecule transport are essential components of technologies that address our pressing need for clean water and energy (e.g., reverse osmosis, batteries, and fuel cells). Designing new polymers with transport properties tailored to emerging separation goals would significantly advance next-generation processes (e.g., lithium extraction and electrochemical CO2 conversion) while improving existing membrane processes. However, many molecular interactions underpinning membrane throughput and separation efficiency remain poorly understood, impeding the development of new materials with desired performance. To address some of these basic knowledge gaps, this presentation will detail fundamental thermodynamic and transport studies in materials used in water and energy applications. A particular point of emphasis will be connecting water sorption with transport properties that quantify membrane performance (e.g., water permeation and ionic conductivity). The first part of this presentation will focus on state-of-the-art ion exchange membranes and will demonstrate that hydraulic water transport is governed by a solution-diffusion mechanism rather than a pore-flow mechanism. These results will be applied to show that the relationship between water activity and water concentration (i.e., sorption) can be used to predict water flux as a function of transmembrane pressure, provided a reasonable estimate of the water diffusion coefficient. The utility of thermodynamic models to predict the experimental data without adjustable parameters, thereby linking performance with basic polymer and solution properties, will be discussed. The second part of this presentation will focus on polyether-based electrolytes relevant to battery applications. Specifically, measurements of ionic conductivity in materials systematically hydrated from dry conditions will be presented. The transition in ion transport mechanisms occurring between rigorously dry and highly swollen polymers, as modulated by water sorption, will be shown. The implications of results from each study on design strategies for materials that exhibit specific separation properties will be addressed.
Enjoy a quick refreshment break.
Abstract:
Svante Technologies Inc. (Svante), a global leader in carbon capture and removal technologies, recognized early the potential of MOFs for CO₂ capture and has since leveraged state-of-the-art laboratory techniques for rigorous material evaluation and screening. Robust characterization capabilities are fundamental to this effort. The ability to accurately assess key sorbent properties from milligram-scale batches is critical to successful development. A combination of standard and customized tools- including single and multicomponent sorption instruments (TGA, DVS and breakthrough systems), PXRD, FTIR, SCXRD, ZLC, CSFR, ESEM/EDS, DMA, and NMR, etc., many with in-situ testing capabilities- enables precise evaluation of kinetic, equilibrium, selectivity, and stability-related properties. These measurements underpin structure–property relationships, guide MOF engineering, and support reliable KPI assessment for final capture units. This approach led to the identification of CALF-20 as a leading MOF for CO₂ capture and eco-friendly synthesis supporting reliable tonne-scale production [1]. CALF-20 is deployed in Svante’s structured sorbent filters, manufactured at its Gigafactory using automated processes and integrated with the RTSA platform, where they have demonstrated consistent KPI performance in major pilot projects. More recently, Svante’s acquisition of Carbon Alpha has expanded its capabilities into CO₂ transport and geological storage, strengthening its position as a fully integrated carbon management provider across the CCS value chain [3]. Looking ahead, Svante is advancing next-generation stable MOF sorbents with enhanced CO₂ uptake and improved selectivity under low CO₂ concentrations and high humidity conditions.
References:
[1] JB Lin, T. Nguyen, et al. Science, 2021. 374 (6574), 1464-1469.
Panel discussion with special guest Dr. Lisa Sun, Assistant Professor at University of Birmingham and SMS scientists.
Topics: TBD
May 20th, 2026: Day 2 Schedule
A welcome from Dr. Daniel Burnett, Vice President at Surface Measurement Systems to kick off day 2.
Abstract:
Herein, the effects of different conditions on the enthalpy of adsorption are investigated to identify a new method to analyze the carbon black surface. The developed method was then applied to different surface treated carbon blacks.
Abstract:
Surface energy is an important, yet under characterized property in metal combustion studies. The concept of surface energy is discussed in combustion literature, but actual measurements are rare. The Texas Tech Combustion Lab designed and performed a series of experiments using the Surface Measurement System Surface Energy Analyzer to quantify surface energy of aluminum powder. Aluminum powder has a high energy density (i.e., 31 kJ/g) and is commonly used to generate energy from its oxidation reaction. Surface properties influence ignition sensitivity, and smaller particles with higher surface area to volume ratios provide increased contact between fuel and oxidizer particles, thereby inciting ignition more readily. Beyond interfacial contact, surface energy will influence ignition sensitivity. This study examined the influence of particle size on surface energy, with particle diameters ranging from 2-50 micrometers. A general trend was found that the smaller particles have higher and more homogeneous surface energy distribution. Analysis was extended to thermally treated aluminum particles, which were annealed and quenched to induce internal stress and improve the reaction performance. Results show a newfound trend, where the surface energy increased in thermally treated relative to untreated particles that were above a threshold size, but reduced in particles below the threshold. The threshold size also corresponds to a shift in reaction mechanism from diffusion to kinetically controlled. Vapor phase combustion describes diffusion-controlled reactions whereas the flame approaches the surface of the particle and multi-phase reactions ensue for kinetically controlled reactions. The results suggest that thermal treatment has a different effect on smaller compared with larger particles, but in both cases the effect promotes reactivity according to the shift in mechanistic oxidation process.
Enjoy a quick refreshment break.
Abstract:
Solute-solvent interactions between a commercial soya-based lecithin and its molecular constituents are relatively limited in the literature and critical to understanding the miscibility and solubility of lecithin in various solvents. To better understand lecithin’s solubility characteristics in various solvents, an inverse gas chromatographic (IGC) study was conducted, utilizing a commercial lecithin (68% phospholipids) as a column substrate and 28 solute probes that vary with respect to their volatility and polarity over the temperature range from 55-100oC. Precise determination of the solute retention volume data on the lecithin columns yielded activity coefficients, Henry’s Law constants, and chi interaction parameters for the various solute probe – lecithin pairs, and solubility parameters for lecithin as a function of temperature. The log of the specific retention volume vs. reciprocal of temperature plots were linear and varied less than 5% between multiple lecithin columns. Most of solute probes at conditions approaching infinite dilution have χ parameters less than their corresponding χc values, indicating miscibility and solubility in lecithin. Lecithin’s solubility parameter (δ) was found to vary from 20.0 to 18.3 MPa1/2 from 55 to 100oC, respectively, in reasonable agreement with those calculated from group contribution method based on the molecular composition of the studied lecithin. Similar data from IGC experiments were also determined using a highly purified PC (phosphatidylcholine) column in triplicate for comparison with the above lecithin values. This included mole and weight fraction activity coefficients, Henry’s Law and partition coefficients, interaction and solubility parameters, as well as heats of solution and mixing. Mole fraction activity coefficients of the organic solute probes in PC show both positive and negative deviations from Raoult’s Law. Activity coefficients less than unity are exhibited by both aromatic and chlorinated classes of solutes and particularly ethanol – all solute activity coefficients showing the expected trend with increasing temperature. The experimental determined solubility parameters (δ) for PC over the above-stated temperature range vary from 20.5 – 18.3 MPa1/2, δ data which has not been available previously. Enthalpy data, i.e., heats of solution and mixing indicate that at this concentration, that the recorded heats of solution are dominated by the heats of solute vaporization. The utilization of this IGC-derived data base will be discussed with respect to using it in the extraction, dissolution, and purification of phospholipids- and related lipid-based excipients and there use in drug formulation delivery systems, i.e., liposomes.
Abstract:
One of the largest issues facing sorption science in the areas of soft matter, such as polymers and coatings, is maintaining surface area with temperature. These materials are also difficult to measure using traditional methods such as contact angle, as these materials possess semicrystalline nature or solubility issues. Additionally, the time scale of a typical iGC experiment necessarily leads to flow in traditional sample geometries combined with phase changes, such as observed for polymers above the glass transition and melt temperatures. To address this issue, we have designed a sample cell that can accommodate solid supported films that mitigate some of these issues. Our initial efforts have been in the areas of reverse osmosis membranes and thermoplastics. We will present our findings and how they are connected to manufacturing and industrial needs.
Enjoy lunch and also tour the US Forest Products Laboratory in small groups.
Abstract:
Spray drying is widely used to engineer respirable pharmaceutical powders, yet predicting aerosol performance from formulation composition remains challenging. While bulk properties such as glass transition temperature and crystallinity are routinely characterized, they often fail to explain variability in powder dispersibility. Surface energetics — particularly their sensitivity to environmental humidity — may offer a more mechanistic link between formulation design and real-world aerosolization behavior.
This presentation explores the application of inverse gas chromatography with surface energy analysis (IGC-SEA) to characterize the surface properties of spray-dried pharmaceutical powders spanning distinct formulation classes: a ternary carbohydrate–protein system (lysozyme with trehalose, inulin, and mannitol), a combination antibiotic dry powder (clofazimine–amikacin), and an amorphous solid dispersion (vonoprazan–PVP). Across these systems, IGC-SEA was used to measure dispersive and specific surface energy components, Lewis acid–base character, and humidity-dependent surface reorganization. By systematically varying relative humidity during probe injections, we identified formulation-specific thresholds at which surface basicity changes, reflecting moisture-induced restructuring of the outermost molecular layers. In the protein–carbohydrate system, the humidity threshold for surface reorganization was strongly correlated with aerosol dispersibility, suggesting that dynamic surface moisture sensitivity — rather than static surface energy — governs interparticle cohesion. Analogous surface energetic signatures were observed in the clofazimine–amikacin and vonoprazan–PVP systems, where IGC-SEA provided insight into component surface enrichment and its consequences for powder handling and aerosolization. These case studies collectively demonstrate that IGC-SEA, particularly under controlled humidity conditions, serves as a powerful and underutilized tool for linking spray-drying process outcomes to functional powder performance across diverse pharmaceutical applications.
Abstract:
The development of robust pharmaceutical drug products requires a clear understanding of how crystal structure, moisture interactions, and particle properties collectively influence performance. Although crystal engineering and particle engineering are often treated independently, practical challenges necessitate an integrated approach.
An integrated crystal and particle engineering framework is described, incorporating solid form selection (polymorphs, salts, and co-crystals) and solid-state characterization using X-ray powder diffraction (XRPD), differential scanning calorimetry (DSC), and thermogravimetric analysis (TGA). Particular emphasis is placed on moisture–solid interactions, evaluated through dynamic vapor sorption (DVS), to understand hygroscopicity, stability, and moisture-induced transformations.
Case studies, including a 5-fluorocytosine-based system, demonstrate how moisture sorption analysis identifies stability limitations and informs solid-form selection. The relationship between crystal structure and mechanical properties, such as tabletability, is examined, along with strategies to improve performance through crystal engineering. The translation of molecular-level insights into particle-level attributes—such as particle size, flowability, and compressibility—is further explored through particle characterization and formulation studies.
The integration of moisture sorption, solid-state characterization, and particle engineering provides a practical pathway to address challenges related to physical stability, mechanical properties, and manufacturability in drug product development.
Abstract:
Poly(lactic acid) [PLA] is increasingly used in packaging, but contact with alcoholic foods can alter its structure and affect the release of low–molecular–weight species. A key indicator of these changes is the relationship between the exposure temperature and the in situ glass transition temperature measured in the contacting medium (Tg-in situ), expressed as ΔT = T - Tg-in situ. In this study, PLA films were immersed in ethanol (EtOH) – water food simulants containing 10%, 50%, or 100% (v/v) ethanol at temperatures below and above Tg-in situ. Lactic acid (LA) oligomers migrating into the liquids were quantified by UPLC–MS/MS over time. At all temperatures, the monomers released into 100% ethanol were lower than those from solutions with lower ethanol concentrations at the same immersion time. Temperature effects aligned with ΔT: at 20 °C (ΔT = −4.3 °C), LA-1 release was initially lower than at 40 °C (ΔT = 15.7 °C), but both conditions approached similar levels after 30 days. At 60 °C (ΔT in 10% EtOH = 8.1 °C, in 50% EtOH = 23 °C), the amount of LA-1 released was significantly higher in the 10% and 50% ethanol solutions compared to the 40°C and 20°C conditions. Additionally, a higher LA-1 release was observed in the 50% solution on day 10. However, by day 30, the 10% solution showed greater LA-1 release. Under higher-ΔT conditions in 50% ethanol, oligomers as large as LA-16 were detected after 10 days. After 30 days, the largest oligomer species decreased to LA-12, indicating ongoing hydrolysis. Overall, the data demonstrate that ethanol concentration, contact duration, and temperature relative to Tg-in situ, collectively influence both the extent of release and the distribution of oligomers. These findings offer a practical basis for assessing PLA performance and potential migrants under realistic time–temperature conditions relevant to alcoholic media in food and medical packaging.
Enjoy a quick refreshment break.
Abstract:
Evaporative water loss from soft contact lenses is an important factor leading to contact lens induced dry eye (CLIDE) and is important to the understanding of end of day comfort. This presentation describes a new method for ranking the moisture retention properties of soft contact lenses. The method ranks the lenses according to their moisture retention rate. Experiments were carried out using a dynamic vapor sorption instrument (DVS) to measure the evaporation rate of water from pure saline (no contact lens) and the evaporation of water through a contact lens. The moisture retention rate is defined as the reduction in the evaporation rate, relative to saline, caused by the presence of the contact lens. The final scale for ranking moisture retention was converted to a percentage scale (0-100% moisture retention) and is referred to as a moisture retention index. Moisture retention rates across the 20 commercially available contact lens materials studied ranged from 6% to 72%. The experimental details of these measurements will be discussed along with trends in moisture retention that may be related to the composition of the material and other factors such as porosity.
Panel discussion with SMS scientists. Topics: TBD
Hear from Leaders in Sorption and Surface Science
The symposium will host invited talks from prominent academic researchers, government scientists, and industry experts. These speakers represent diverse perspectives within the sorption science community, spanning material characterization, adsorption thermodynamics, and applied surface analysis.
Discover the Current Speaker List

Dr. Ashlee Brunaugh
Assistant Professor, Pharmaceutical Sciences at University of Michigan

Dr. Brianna Coia
Product Scientist at Birla Carbon

Cason Jones
Graduate Research Assistant at Texas Tech University

Cory Sauer
Application Engineering Specialist at 3M

Dr. Jamie M. Kropka
Technical Staff at Sandia National Laboratories

Dr. Jeremiah Woodcock
Research Chemist at NIST

Dr. Jerry King
R&D Consultant/Educator at CFS (Critical Fluid Symposia)

Dr. Nima Masoumifard
Director, MOF & Characterizations at Svante

Dr. Rahul Sujanani
Assistant Professor of Chemical and Biological Engineering at University of Wisconsin-Madison

Dr. Rafael Auras
Professor at Michigan State University

Dr. Sathyanarayana Reddy Perumalla
Founder and Director, Solid State Chemistry and Analytical Services at Seven Star Pharmaceutical Services

Dr. Sam Zelinka
Project Leader, Building and Fire Sciences at US Forest Service

Dr. Venkateswara Rao Kode
Assistant Professor of Chemical Engineering at University of Tennessee at Chattanooga

Dr. Carlos D. Díaz-Marín
Assistant Professor of Energy Science and Engineering at Stanford University

Dr. David Keisar
Postdoctoral Associate at Massachusetts Institute of Technology

Dr. Donald Riederer
Principal Scientist at Johnson & Johnson Vision Care, Inc.

Dr. Ipsita Mishra
Solids Processing/Particle Technology SME at Corteva Agriscience
Venue and Travel
The 2026 North American Sorption Symposium will take place at the USDA Forest Products Laboratory, located on the University of Wisconsin–Madison campus. This historic facility is a leading center for materials and sustainability research, providing an ideal setting for scientific collaboration and discussion.
Madison is easily accessible via Dane County Regional Airport (MSN), located about 20 minutes from the venue. For those driving, parking is available both at the hotel and the USDA Forest Products Laboratory.
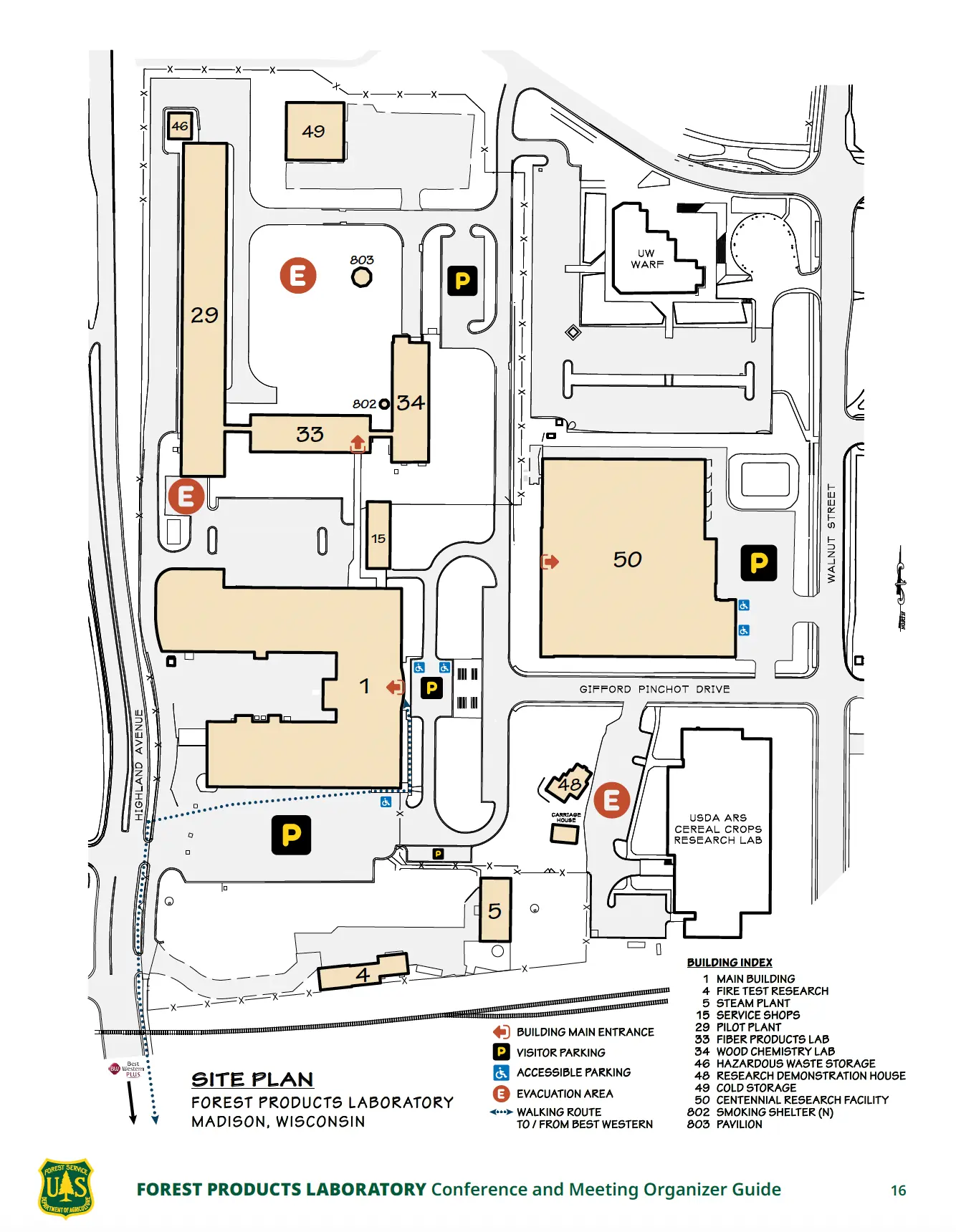
Venue Details:
What's the Address?
- 1 Gifford Pinchot Drive Madison, WI 53726
- Building #1 Room 500
Parking at the Products Forestry Laboratory
There are several free parking areas near the event. The main entrance parking for Building #1 can be accessed off Walnut Street. The South parking lot off Highland Avenue is fenced. It is available for foot traffic and can be walked through to reach the front entrance of Building #1.

Hotel Accommodations:
A room block has been reserved for symposium attendees at:
Best Western Plus InnTowner Madison
2424 University Avenue | Madison, WI 53726
- Just an 11-minute walk or 3-minute drive from the venue
- Conveniently located near restaurants, coffee shops, and the UW–Madison campus
Reserve Your Spot at the 2026 Sorption Symposium
Registration is open. Space is limited, and early registration is recommended to secure your place among the leading voices in sorption research.
Lunch will be provided both days.
Early Bird
Rates (Through January 31st)
-
Delegate Pass
$385(Sold Out)
-
Student Pass
$125(Sold Out)

Standard
Rates
-
Delegate Pass
$445
-
Student Pass
$155
Call for Posters
Highlight your research at the 2026 Sorption Symposium!
Our poster session offers an excellent opportunity for students and researchers to present new findings, exchange ideas, and connect with fellow attendees.
To submit a poster, indicate your interest when completing registration.
Submission Deadline: March 20, 2026

